Clinical Trials’ Bottlenecks as Critical Impediment to Translational Medicine
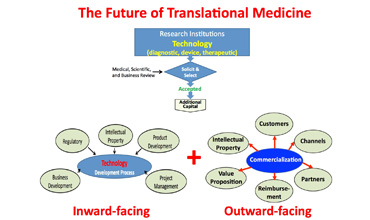
Abstract:
Translational
medicine has made impact and have come to be accepted (Dai, et al., 2013) as a
global medical strategy for improving Medicare such that, funding of
translational medicine globally has increased, it is treated with priority and
many institutes for translational studies have been established, (Zhang, 2012).
Demonstrating this critical significance of translational medicine to modern
medical practice is the emphasis by the World Medical Association (WMA Ethics Manual, 2009), that ‘physicians must know how to interpret
research results and apply them to patients’.
However,
as noted by Zerhouni, (2007) and Qian et al., (2012), a lot remains to be
understood and lessons need to be learned on the best strategies for translational
medicine which may not be unconnected with observed issues of clinical research
bottlenecks. Mankoff, et al., (2004), had earlier identified “three major
obstacles to effective translational medicine”. The first two revolves around
the subject of clinical research; – “the challenge of translating basic science
discoveries into clinical studies”, and “the translation of clinical studies
into medical practice and health care policy”. This second issues hinges on the
core challenges of the practical conduct of clinical research. Sung, et al.,
(2003) opined that the two
“translational blocks can be removed only by the collaborative efforts of
multiple system stakeholders”
Essential for research translation is data
generated from clinical research. Clinical trials operations require
cooperation among a diverse group of stakeholders including research sponsors
(industry, academia, government, non-profit organizations, and patient
advocates), clinical investigators, patients, funders, physicians, and regulators.
This was aptly captured by Dai,
et al., (2013), who noted that “enormous collaborative and multidisciplinary work is
required prior to when the results of scientific research can be translated
into effective clinical practice”. Each stakeholder offers
a different set of tools to support the essential components of a clinical
trial. These resources form the infrastructure that currently supports clinical
research (English,
et al., 2010). Time, money, personnel, materials (e.g., medical
supplies), support systems (ICT as well as manpower), and a clear plan for
completing the necessary steps in a trial are all part of the clinical research
infrastructure.
Significant time, energy, and money from the
different collaborators are spent on bringing the disparate resources together.
Because these resources are not streamlined, clinical trials infrastructure
pose challenges to investigators working on new research questions and most
times must start afresh without drawing on existing resources. This imminent
lack of harmony and disparate clinical research resources imposes bottlenecks
that constitute impediments on the planning, execution and outcome of research
which by implication either slow down or prevent translation outright.
References:
1. 45
CFR §46.111(a)(4).
2. Aban, I. B., Wolfe, G. I.
Cutter, G. R., Kaminski, H. J. Jaretzki
III A., Minisman, G., Conwit, R., and Newsom-Davis, J., [on behalf of MGTX Advisory Committee], (2008). The MGTX Experience: Challenges in Planning
and Executing an International,
Multicenter Clinical Trial J
Neuroimmunol.; 201-202: 80–84. doi:10.1016/j.jneuroim.2008.05.031.
3.
Beauchamp, T.L. and
Childress J. (1996). The Principles of
Biomedical Ethics. New York, NY: Oxford University Press;
4. Califf,
R. M. 2009. ACS and Acute heart Failure Models. Speaker presentation at the
Institute of Medicine Workshop on Transforming Clinical Research in the United
States, October 7–8, 2009, Washington, DC
5. Dai,
K-R., Yang, F., Gan, Y-K., (2013). Development of translational medicine in
China: Foam or feast? Journal of Orthopaedic Translation, 1, 6-10
6. Di
Masi, J. A., R. W. Hansen, and H. G. Grabowski. 2003. The price of innovation:
New estimates of drug development costs. Journal
of Health Economics 22(2):151−185.
7. English,
R., Lebovitz, Y., and Griffin; R., (2010).Transforming Clinical Research in the United States: Challenges and
Opportunities: Workshop Summary Forum on Drug Discovery, Development,
and Translation; Institute of Medicine
8. IOM.
(2007). Learning What Works best: The Nation’s Need for Evidence on Comparative
Effectiveness in HealthCare. http://www.iom.edu/~/media/Files/Activity%20Files/Quality/VSRT/ComparativeEffectivenessWhitePaperF.ashx.
9.
Lane, H. C. 2009. Improving the Effectiveness and Efficiency
of NIH Intramural Clinical Research. Speaker presentation at the
Institute of Medicine Workshop on Transforming Clinical Research in the United States, October 7–8, 2009,
Washington, DC.
10. Mankoff
S. P., Brander, C., Ferrone, S., and Marincola, F. M., (2004). Lost in Translation: Obstacles to
Translational Medicine Journal
of Translational Medicine 2:14 http://www.translational-medicine.com/content/2/1/14
11. McGlynn,
E. A., S. M. Asch, J. Adams, J. Keesey, J. Hicks, A. DeCristofaro, and E. A.
Kerr. 2003. The quality of health care delivered to adults in the United
States. New England Journal of Medicine 348(26):2635−2645.
12. Qian,
M., Wu, D., Wang, E., Marincola, F., M., Wang, W., Rhodes, W., Liebman, M., Bai,
C., Lam, Ching-Wan, Marko-Varga G., Fehniger T. E., Andersson, R., and Wang,
X., (2012). Development and promotion in translational medicine: perspectives
from 2012 sino-american symposium on clinical and translational medicine;
Clinical and Translational Medicine, 1:25. http://www.clintransmed.com/content/1/1/25
13. Sun, X. G., Lin,
D., Wu, W., Lv, Z., (2011). Translational Chinese Medicine: A Way for
Development of Traditional Chinese Medicine. Chinese Medicine, 2, 186-190 doi:10.4236/cm.2011.24029 (http://www.SciRP.org/journal/cm)
14. Sung,
N.S., Crowley, W.F. Jr, Genel, M., Salber, P., Sandy, L., Sherwood, L.M., et al.: (2003).Central challenges facing the national
clinical research enterprise. JAMA; 289:1278-1287
15. The Belmont Report: Ethical Principles and
Guidelines for the Protection of Human Subjects of Research.
National Commission for the Protection of Human Subjects of Biomedical and
Behavioral Research. Washington, DC: US Government Printing Office; 1979.
16. The Nuremberg Code. JAMA. 1996;276:1691
17.
World
Medical Association Medical Ethics
Manual, 2nd edition (2009)
18. Zhang
J M. (2012). Translational medicine in China. Sci China Life Sci, 55: 834–836,
doi: 10.1007/s11427-012-4373-0
19. Zerhouni,
E. A., (2007). Translational research: moving discovery to practice. Clin.
Pharmacol Ther 81(1):126–128.

